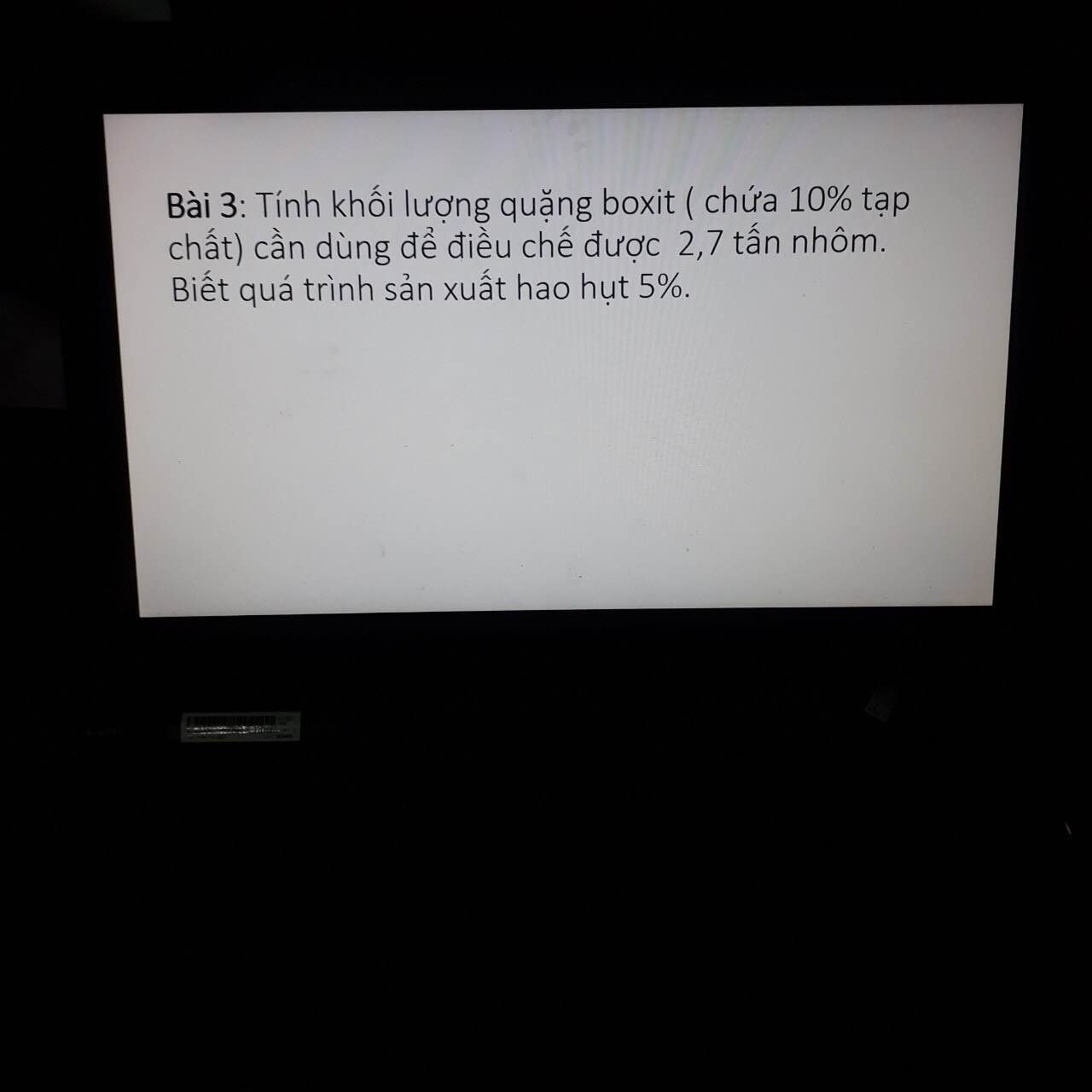
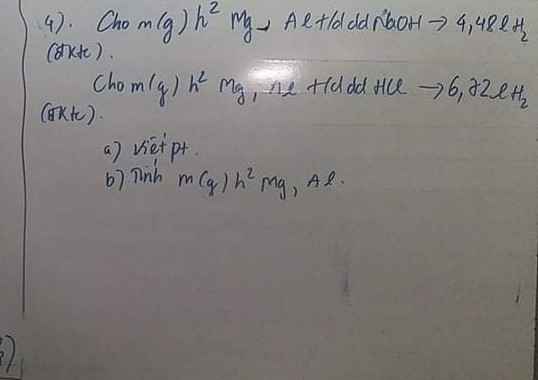\(a.n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\\ Zn+2HCl\rightarrow ZnCl_2+H_2\\ b.0,1........0,2..........0,1.........0,1\left(mol\right)\\ C\%_{ddH_2SO_4}=\dfrac{0,1.98}{140}.100=7\%\\ c.m_{dd.muối}=6,5+140-0,1.2=146,3\left(g\right)\\ C\%_{ddZnCl_2}=\dfrac{136.0,1}{146,3}.100\approx9,296\%\\ \)
a.nZn=6,565=0,1(mol)Zn+2HCl→ZnCl2+H2b.0,1........0,2..........0,1.........0,1(mol)C%ddH2SO4=0,1.98140.100=7%c.mdd.muối=6,5+140−0,1.2=146,3(g)C%ddZnCl2=136.0,1146,3.100≈9,296%